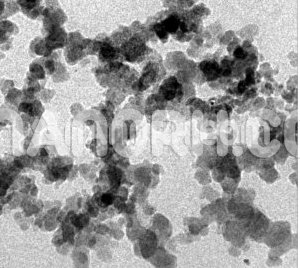
| Iridium Nanoparticles | |
| Product No | NRE-1020 |
| CAS No. | 7439-88-5 |
| Formula | Ir |
| APS | <100nm (Can be Customized) |
| Purity | 99.9% |
| Color | silvery-white |
| Molecular Weight | 192.22 g/ mol |
| Density | 22.56g/cm3 |
| Melting Point | 2446 °C |
| Boiling Point | 4130 °C |
Iridium Nanoparticles
Iridium nanoparticles have a range of applications across various fields due to their unique properties. Iridium is a rare and precious metal that is highly corrosion-resistant and has exceptional catalytic and electrical conductivity properties. When reduced to nanoparticles, these properties become even more pronounced, making them valuable in several areas:
Catalysis: Iridium nanoparticles are used as catalysts in various chemical reactions, including hydrogenation, oxidation, and reduction reactions. Their high catalytic activity and stability make them essential in industries such as petrochemicals, pharmaceuticals, and organic synthesis.
Fuel Cells: Iridium nanoparticles are used as electrocatalysts in fuel cells, which convert chemical energy (usually hydrogen) into electrical energy. Their high catalytic activity helps improve the efficiency and performance of fuel cells.
Electronics: Iridium nanoparticles are employed in the manufacture of high-performance electronic devices, such as organic light-emitting diodes (OLEDs) and organic photovoltaic cells. They enhance the conductivity of materials and improve the efficiency of these devices.
Medicine: Iridium nanoparticles are being explored for various medical applications. They can be used as contrast agents in medical imaging techniques like computed tomography (CT) scans and magnetic resonance imaging (MRI). Additionally, researchers are investigating their potential in targeted drug delivery systems.
Environmental Remediation: Iridium nanoparticles can be used in environmental applications, such as water purification and air pollution control. They can help catalyze the breakdown of pollutants and enhance the efficiency of these processes.
Aerospace and Space Exploration: Iridium nanoparticles are used in aerospace applications, including the manufacture of high-temperature-resistant materials for spacecraft and propulsion systems. Iridium-coated components are crucial for withstanding the extreme conditions of space travel.
Electroplating: Iridium nanoparticles are used in electroplating processes to create highly durable and corrosion-resistant coatings on various materials, particularly for components exposed to harsh environments.
Analytical Chemistry: Iridium nanopowder is utilized in analytical chemistry for the detection of trace elements and compounds in various analytical techniques, including mass spectrometry.
Energy Storage: Iridium nanopowder is being researched for its potential use in advanced energy storage systems, such as supercapacitors, due to their high electrical conductivity and stability.
Cancer Therapy: In research settings, iridium nanopowder is being explored for its potential in cancer therapy. They can be functionalized and used for targeted drug delivery to cancer cells, offering a way to improve the effectiveness of cancer treatments while minimizing damage to healthy cells.