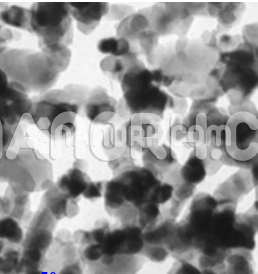
| Manganese Dioxide Nanoparticles | |
| Product No | NRE-3038 |
| CAS No. | 1313-13-9 |
| Formula | MnO2 |
| APS | <100nm (Can be Customized) |
| Purity | 99.9% |
| Color | black |
| Molecular Weight | 86.94 g/mol |
| Density | 5.02 g/cm3 |
| Melting Point | 535 °C |
| Boiling Point | NA |
Manganese DioxideNanoparticles
Manganese dioxide nanoparticles are attracting the attention of nanotechnology researchers due to their low toxicity. Manganese dioxide nanoparticles exist in different crystal structures, with the same basic MnO6 structure, bonded differently. The Manganese Dioxide Nanoparticles can be differentiated based on the bonds in the structure. Both top-down and bottom-up methods can be used to prepare NP from MnO2. Due to the high cost of preparation and structural defects of the NPs produced, the top-down approach is not widely used. Most researchers prefer the bottom-up approach, as particles of uniform size and morphology can be achieved. The wet chemical pathway is used to synthesize NPs from MnO2. This review mainly focuses on the synthesis of widely used wet chemical methods, such as hydrothermal, redox process, sol-gel method, thermal reflux process, chemical precipitation method and green synthesis method. While these methods are widely used, none of them are perfect. Studies are underway to optimize the methods by modifying the reaction parameters or by incorporating microwaves, ultrasonic waves, etc. in the synthesis. NP MnO2 are easy to prepare and have good stability, so they have been applied in various fields. A-MnO2 tunneling was used as an electrocatalyst for oxygen reduction and oxygen evolution in aqueous and organic electrolytes. While layered birnesite has an application as a capacitor. Likewise, along with it, other forms have also been used as a catalyst and adsorbent. They are also used in the field of biomedicine and MnO2 activity has been enhanced by doping the nanomaterials with other metals such as Ni, Ag, etc.