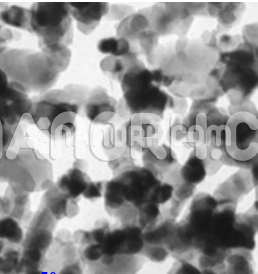
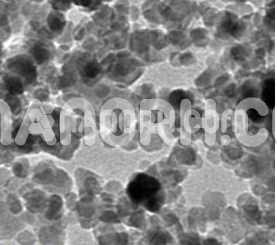
Nickel Iron Oxide (NiFe2O4) Nanopowder / Nanoparticles (NiFe2O4, 99.99%, 20 nm)
Loathsome suspension (for suspended railroad frameworks); High-thickness attractive chronicle media; Magnetic refrigeration; Catalysts; Magnetic fluids; Microwave safeguards.
| Nickel Iron Oxide Nanoparticles | |
| Product No | NRE-4011 |
| CAS No. | 12168-54-6 |
| Formula | NiFe2O4 |
| APS | <100nm (Can be Customized) |
| Purity | 99.99% |
| Color | brown |
| Molecular Weight | 234.381 g/mol |
| Density | 5.368 g/cm3 |
| Melting Point | 1,955 °C |
| Boiling Point | NA |
Nickel Iron Oxide Nanoparticles (NiFe2O4) Application:
Nickel-iron oxide nanoparticles are one of the most important soft materials due to their unique electrical and magnetic properties and their high magnetocrystalline anisotropy, high electrical resistance, chemical stability, and mechanical hardness. For these reasons, this material is used for magnetic memory devices and magneto-optical devices, for cooling, and as sensors. Nickel-iron oxide nanoparticles and NiFe2O4/chitosan compounds show excellent response to glucose electro-oxidation. Conventional methods for producing nickel nanoferrites include mechanosynthesis, coprecipitation, sol-gel, and solvothermal methods. A literature review shows that the magnetic behavior of these nanoparticles mainly depends on the synthesis route. A new synthesis method has recently been developed in our laboratory for the preparation of fairly uniform magnetic nanoparticles with dimensions around 20-50 nm based on the electrochemical oxidation of a metal anode. The final product requires no further purification or secondary size selection process. This article reports on the use of this new methodology to obtain nickel ferrite nanoparticles by electrochemical oxidation of two electrodes, iron and nickel in a solution containing a surfactant to prevent agglomeration of nanoparticles. In addition, by controlling process parameters such as current density, temperature, stirring speed, and surfactant concentration, nickel ferrites of a certain shape and size can be produced. The nanoparticles produced by this process were structurally characterized by X-ray diffraction (XRD). Inductively coupled plasma mass spectrometry (ICP-MS) was used to determine the Fe: Ni ratio in nanoparticles. The microstructure and magnetic properties were determined using transmission electron microscopy (TEM) and vibrational sample magnetometry (VSM) techniques. Finally, a catalyst electrode for glucose oxidation with nickel ferrite nanoparticles was prepared.