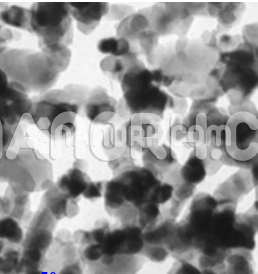
| Tellurium Nanopowder / Nanoparticles | |
| Product No | NRE-1044 |
| CAS No. | 13494-80-9 |
| Formula | Te |
| APS | <100nm (Can be Customized) |
| Purity | 99.9% |
| Color | Gray |
| Molecular Weight | 127.60 g/mol |
| Density | 6.25 g/cm³ |
| Melting Point | 449.5 °C |
| Boiling Point | 988 °C |
Tellurium Nanopowder / Nanoparticles
Tellurium nanoparticles have gained attention in various fields due to their unique properties and potential applications. Some of the notable applications of tellurium nanoparticles include:
Thermoelectric Materials: Tellurium nanoparticles are used in thermoelectric materials to convert heat into electricity. They have high thermoelectric efficiency, making them valuable for applications in waste heat recovery and power generation in remote locations.
Photovoltaics: Tellurium nanoparticles are used in thin-film photovoltaic solar cells. Tellurium-based solar cells have the advantage of being less expensive to produce compared to traditional silicon solar cells.
Semiconductor Industry: Tellurium nanoparticles are used in the semiconductor industry for the production of electronic devices such as transistors and photodetectors. They can be employed to create nanoscale features in electronic components.
Optoelectronics: Tellurium nanoparticles are utilized in optoelectronic devices, including infrared detectors and sensors. Their unique optical properties make them suitable for various applications in sensing and imaging.
Catalysis: Tellurium nanoparticles have been investigated as catalysts in chemical reactions. They can facilitate various catalytic processes, including reduction and oxidation reactions, due to their high surface area and reactivity.
Medical Imaging: Tellurium nanoparticles can be used as contrast agents in medical imaging techniques like X-ray and computed tomography (CT) scans. They enhance the visibility of specific tissues or organs in diagnostic imaging.
Drug Delivery: Tellurium nanoparticles can serve as drug carriers for targeted drug delivery. Their surface properties can be modified to attach specific drugs or therapeutic agents, allowing for precise drug delivery to the desired site.
Nanotechnology Research: Tellurium nanoparticles are essential tools in nanotechnology research. They are used as building blocks for the development of nanoscale structures and devices, including nanowires, nanotubes, and nanosensors.
Biological and Environmental Sensing: Tellurium nanoparticles can be functionalized to detect various biological and environmental analytes, including heavy metals, toxins, and biomolecules. They are used in biosensors and environmental monitoring systems.
Energy Storage: Tellurium nanoparticles are being explored for their potential in energy storage systems, such as rechargeable batteries and supercapacitors, where they can improve the energy density and performance of these devices.