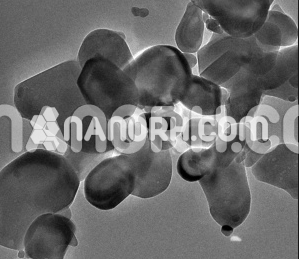
Terbium Oxide (Tb4O7) Nanopowder / Nanoparticles (Tb4O7, 99.99%, 10-100 nm, Cubic)
Terbium(III,IV) oxide, occasionally called tetraterbium heptaoxide, has the formula Tb4O7. Tb4O7 is one of the main commercial terbium compounds, and the only such product containing at least some Tb(IV) (terbium in the +4 oxidation state), along with the more stable Tb(III). It is produced by heating the metal oxalate, and it is used in the preparation of other terbium compounds……..
| Terbium Oxide Nanopowder | |
| Product No | NRE-3059 |
| CAS No. | 12037-01-3 |
| Formula | Tb4O7 |
| APS | <100nm (Can be Customized) |
| Purity | 99.99% |
| Color | Dark brown-black |
| Molecular Weight | 747.70 g/mol |
| Density | 7.3g/cm3 |
| Melting Point | 2,410 °C |
| Boiling Point | 3123°C |
Terbium Oxide Nanopowder Tb4O7 Application:
Terbium(III, IV) oxide, occasionally called tetraterbium heptaoxide, has the formula Tb4O7. Tb4O7 is one of the main commercial terbium compounds, and the only such product containing at least some Tb(IV) (terbium in the +4 oxidation state), along with the more stable Tb(III). It is produced by heating the metal oxalate, and it is used in the preparation of other terbium compounds. Terbium forms three other major oxides: Tb2O3, TbO2, and Tb6O11. Main use: Activating agents for fluorescent powders; CeO2-Tb4O7 composites for catalytic automobile exhaust converters; Glass materials with a Faraday Rotation effect for optical and laser-based devices; Magneto-optical glasses; Magneto-optical recording materials. Terbium oxide nanopowders whose phase transformations were investigated by methods of high-temperature differential scanning calorimetry, dilatometry, and Xray diffraction analysis. The as-prepared powders consisting of Tb7O12 and Tb11O20 phases were converted to Tb2O3 after calcination at 600-800 °C in reducing atmosphere. The sintering behavior of Tb2O3 was studied under microwave heating up to 1780 °C. The microstructure of the powders and ceramics was investigated by scanning electron microscopy. Near full-density material was obtained at about 1620 °C. Further temperature increases causes a deterioration of the ceramics microstructure due to monoclinic Tb2O3 phase formation.