| VANADIUM CARBIDE NANOPOWDER | |
| Product No | NRE-5250 |
| CAS | 12070-10-9 |
| Purity | 99.9% |
| APS | <100 nm (can be customized) |
| Formula | VC |
| Molecular Weight | 62.95 g/mol |
| Density | 5.77 g/cm³ |
| Color | Black |
| Melting Point | 2810 °C |
| Boiling Point | NA |
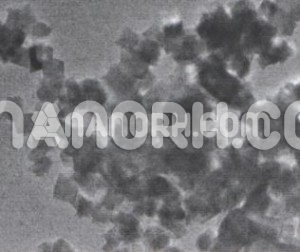
VANADIUM CARBIDE NANOPOWDER
VC Nanopowder were investigated, including direct element reaction, mechanical alloy, programmed temperature reaction, and gas-carburization reduction. However, industrial applications of the methods are still limited due to agglomeration problems, large size distributions, low yields, complex monitoring and high costs. Some newer methods, including the pyrolysis of precursors derived from polymers, sol-gels or solutions, can be used to synthesize sub-micron- or nano-sized carbide particles for ceramics. Such methods have a number of potential advantages: (i) high homogeneity of the reactants can be achieved, (ii) the composition required for ceramic formation can be adjusted by solution processes, and (iii) the polymer or precursor can be subjected to low temperature. Decomposition, allowing the formation of the ceramic in milder conditions than those used in conventional techniques. Examples of elements of group VIb are chromium salicylate pyrolysis, electrochemically prepared chromium polymer precursors and solution derived molybdenum and tungsten precursors. In this experiment, the solution-derived precursor method was used to synthesize VC nanopowder. As we know, it has rarely been reported that VC Nanopowder were synthesized by the solution-derived precursor method using ammonium vanadate (NH4VO3) and nanoscale carbon black as the vanadium source and carbon source, respectively. The present work provides a reference and an attempt for the cost-effective and efficient production of VC Nanopowder for potential industrial applications.