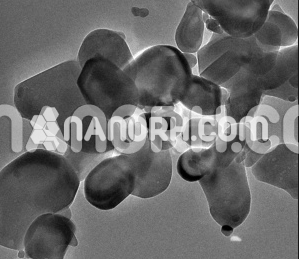
| Ytterbium Nanopowder | |
| Product Number | NRE-1049 |
| CAS No. | 7440-64-4 |
| Formula | Yb |
| Molecular Weight | 173.04 g/mol |
| APS | <100 nm (Can be Customized) |
| Purity | 99.9% |
| Color | Silvery White |
| Density | 6.90 g/cm3 |
| Melting Point | 824 °C |
| Boiling Point | 1,196 °C |
Ytterbium Nanopowder
Ytterbium nanopowder is a form of ytterbium (Yb) that has been processed into very fine particles with nanoscale dimensions. Ytterbium is a chemical element with the atomic number 70 and is part of the lanthanide series on the periodic table. Ytterbium is known for its unique optical and electronic properties, making it valuable in various scientific and technological applications, including:
Lasers: Ytterbium-doped laser materials are widely used in solid-state lasers, particularly in high-power and fiber lasers. Ytterbium-doped lasers have applications in material processing, telecommunications, and medical equipment.
Medical Imaging: Ytterbium nanoparticles can be used as contrast agents in medical imaging techniques like magnetic resonance imaging (MRI) and computed tomography (CT) scans.
Photovoltaics: Ytterbium nanoparticles can be incorporated into solar cell designs to enhance their efficiency in converting sunlight into electricity.
Catalysts: Ytterbium nanoparticles can serve as catalysts in chemical reactions, particularly in the production of biodiesel and other chemical processes.
Scintillation Detectors: Ytterbium-doped nanoparticles can be used in scintillation detectors for measuring ionizing radiation.
Research and Development: Ytterbium nanopowder is utilized in research and development settings to study its unique properties and potential applications in emerging technologies.