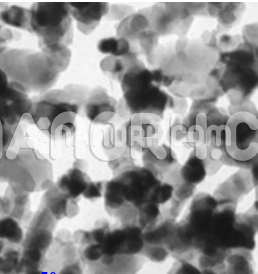
Ag-Cu Alloy Nanopowder/Nanoparticles (<100nm, 99.9%)
- Ag-Cu Alloy Nanopowder/Nanoparticles are to a great extent attention-grabbing because their properties are flexible and can be very distinct from the particles of their constituents. The optical properties of alloy nanoparticles can be different by side effects and composition of their pure components.
- Besides, regularly alloying Ag into Cu to create Ag–Cu alloy nanoparticles could be used to improve the fast oxidizing property of Cu nanoparticles. There are many methods that have been reported on the synthesis of Ag, Cu, and Ag-Cu Alloy Nanopowder/Nanoparticles. As chemical reduction methods for synthesis of those nanoparticles. This technique utilizing different reagents namely metal salt precursors, reducing agents, and stabilizers, as well as their effects on respective nanoparticles, are many important factors. Other parameters such as pH and temperature that have been considered as an important factor influencing the quality of Ag-Cu Alloy Nanopowder/Nanoparticles.
| Ag-Cu Alloy Nanopowder/Nanoparticles | |
| Product No | NRE-2001 |
| CAS No. | 7440-22-4 / 7440-50-8 |
| Formula | AgCu |
| APS | <100nm (Can be Customized) |
| Purity | 99.9% |
| Color | Gray to Black/Brown |
| Molecular Weight | 171.41g/mol |
| Density | 9.7-10.4 g/cm3 |
| Melting Point | 779-900 °C |
| Boiling Point | NA |
Ag-Cu Alloy Nanopowder/Nanoparticles
- Silver (Ag) and copper (Cu) nanoparticles have shown great potential in a variety of applications due to their excellent electrical and thermal properties resulting in high demand in the market. A decrease in size to the nanometer scale has shown a distinct improvement in these inherent properties due to the larger surface-to-volume ratio. Ag and Cu nanoparticles are also shown higher surface reactivity and therefore be used to improve the interfacial and catalytic process. Their melting points have also dramatically decreased compared with bulk and thus can be processed at relatively low temperatures.
- Ag-Cu Alloy Nanopowder/Nanoparticles are significant nanomaterials. Among metal fillers, Ag flakes are most widely used because Ag has the highest electrical conductivity and, unlike many other metals, their oxides are also relatively conductive. However, there have been some concerns about the electro migration of Ag ions in the Ag-filled adhesives under high humidity and electrical bias. Thus, copper has been proposed as an alternative conductive filler in electrically conductive polymer composites for the next generation of lead-free interconnect materials, due to its low cost, high electrical conductivity, low electrical migration, high compatibility with the next generation of Cu interconnect technology. However, due to its easy oxidation nature, the use of pure copper in polymer composites has been limited.