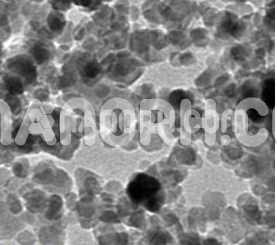
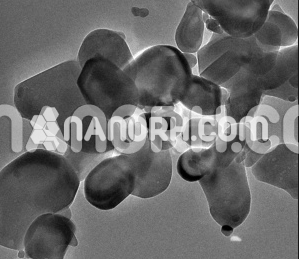
| Chromium Oxide Nanopowder | |
| Product No | NRE-3012 |
| CAS No. | 1308-38-9 |
| Formula | Cr2O3 |
| APS | <60nm (Can be Customized) |
| Purity | 99.9% |
| Color | Green |
| Molecular Weight | 151.990 g/mol |
| Density | 5.22 g/cm3 |
| Melting Point | 2435 °C |
| Boiling Point | 4000 °C |
Chromium Oxide Nanopowder (Cr2O3) Applications:
Chromium(III) oxide is the inorganic compound of the recipe Cr2O3. It is one of the main oxides of chromium and is utilized as a shade. In nature, it happens as an uncommon mineral eskolaite. Due to its extensive strength, chromium is normally utilized in color and was initially called viridian. It is utilized as a part of paints, inks, and glasses. It is the colorant in “chrome green” and “institutional green.” Chromium(III) oxide is the antecedent to the attractive shade chromium dioxide, as indicated by the accompanying response: Cr2O3 + 3 CrO3 → 5 CrO2 + O2. It is one of the materials that are utilized when cleaning the edges of blades on a bit of cowhide (additionally called stropping).Presentation to air, simple to ingest dampness, however does not create water mixes. It is solvent in nitric corrosive. At the point when warmed to over 1200 oC, nano-cobalt oxide will be separated into sub-cobalt oxide. In the hydrogen fire, nano-cobalt oxide is warmed to 900 oC, it will be changed into metal cobalt. Cobalt(II,III) oxide is substance compound with the equation Co3O4.