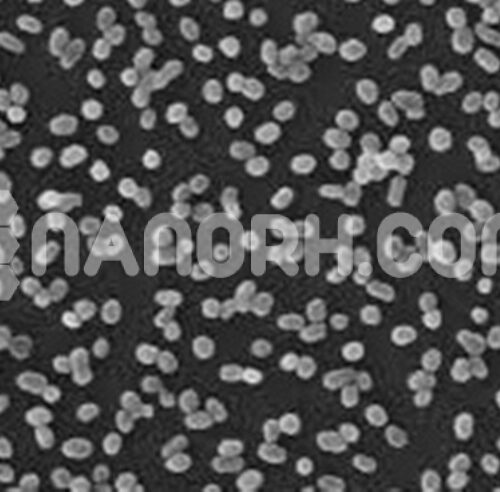
| Graphite Powder | |
| Product No | NRE-8021 |
| CAS No. | 7782-42-5 |
| Formula | C |
| Molecular Weight | 12.01g/mol |
| APS | <10 um (Can be Customized) |
| Purity | 99.95% |
| Color | black |
| Density | 2.2 g/mL at 25 °C |
| Melting Point | 3652-3697 °C(lit.) |
| Boiling Point | 4830°C |
Graphite Powder
Graphite powder has a wide range of applications due to its unique properties, including high electrical and thermal conductivity, lubrication properties, chemical stability, and more. Some common applications of graphite powder include:
Lubrication: Graphite powder is an excellent dry lubricant due to its low friction properties. It is often used in applications where traditional oil-based lubricants are not suitable, such as in high-temperature environments.
Electrical applications: Graphite is used as a conductive material in various electrical components, including electrodes, and brushes, and as a filler in composites to enhance electrical conductivity.
Thermal management: Graphite is used in thermal management applications, such as in heat sinks, thermal pastes, and coatings for electronic devices and high-temperature environments.
Refractories and crucibles: Graphite is used in the production of refractory materials and crucibles, which are essential in high-temperature industrial processes like metal casting and glass manufacturing.
Powder metallurgy: Graphite is used as a mold-release agent and as a powder in the production of metal and ceramic parts through powder metallurgy techniques.
Foundry operations: Graphite is used as a parting agent and mold release agent in foundry operations to help separate castings from molds easily.
Coatings and paints: Graphite can be used as a pigment or filler in various coatings and paints to provide electrical conductivity, corrosion resistance, and lubrication properties.
Friction materials: Graphite is used in the production of friction materials, such as brake linings and clutch plates, to reduce wear and improve performance.
Batteries: Graphite powder is a key component in lithium-ion batteries, where it is used as the anode material due to its ability to store and release lithium ions.
Aerospace and defense: Graphite powder is used in aerospace and defense applications for its lightweight and high-temperature properties. It can be found in aircraft components, missile systems, and rocket nozzles.
Nuclear industry: Graphite is used as a moderator and structural material in certain types of nuclear reactors due to its ability to slow down and control nuclear reactions.
Additive manufacturing (3D printing): Graphite powder can be used as a feedstock material in additive manufacturing processes to create parts with unique properties, such as high thermal conductivity.
Sealing and gasket materials: Graphite powder is often incorporated into gasket and sealing materials to create effective and durable seals in high-temperature and high-pressure environments.
Carbon brushes: Graphite brushes are commonly used in electric motors and generators to provide electrical contact with rotating components, as they have good electrical conductivity and wear resistance.
Art and writing: Graphite powder is used in the production of pencils and drawing materials, and it is also used in various artistic applications.