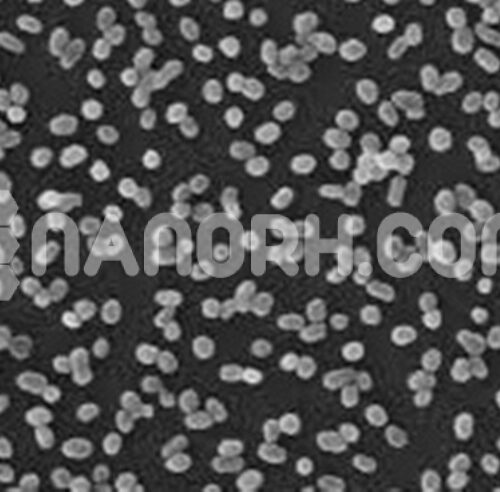
| Niobium Powder | |
| Product No | NRE-8034 |
| CAS No. | 7440-03-1 |
| Formula | Nb |
| Molecular Weight | 92.9g/mol |
| APS | <40um( can be customized) |
| Purity | 99.9% |
| Color | Silver |
| Density | 8.57 g/cm3 |
| Melting Point | 2477 °C |
| Boiling Point | 4744 °C |
Niobium Powder
Niobium powder, derived from the element niobium, has several unique properties that make it valuable in various industries and applications. Some of the key applications of niobium powder include:
Superconductors: Nb powder is commonly used in the production of superconducting materials, which have the ability to conduct electricity with zero resistance. These materials are crucial for applications in medical imaging (MRI machines), particle accelerators, and magnetic resonance systems.
Alloying: Nb powder is often alloyed with other metals, such as steel, to enhance their strength, toughness, and resistance to corrosion. Niobium alloys find use in the aerospace industry for manufacturing aircraft engines, gas turbines, and other high-stress components.
Electronics: Due to its superconducting properties, niobium powder is used in the production of capacitors and other electronic components. Niobium oxide capacitors have superior performance compared to traditional tantalum capacitors, making them valuable in various electronic devices, including smartphones, computers, and other consumer electronics.
Nuclear industries: Nb powder is utilized in the nuclear industry for its resistance to corrosion and its ability to withstand high temperatures. It is used in nuclear reactors and related applications where materials must withstand harsh environments and extreme conditions.
Optics and optics coating: Niobium compounds find applications in the optics industry, where they are used as coatings on lenses and other optical components. These coatings provide improved durability, scratch resistance, and anti-reflective properties.
Medical applications: Niobium powder is used in the medical industry for various applications, including in the production of medical implants, such as orthopedic implants and dental materials. Niobium’s biocompatibility and resistance to corrosion make it suitable for use within the human body.
Chemical processing: Niobium powder is utilized in various chemical processes, including the production of high-performance alloys, superalloys, and catalysts. Its high melting point and resistance to corrosion make it valuable in environments where other materials may degrade.
Other applications: Niobium powder finds use in various other fields, such as in the production of high-strength glass, jewelry, and specialized coatings for industrial equipment.