| Silica Nanoparticles | |
| Product No | NRE-3053 |
| CAS No. | 7631-86-9 |
| Formula | SiO2 |
| APS | <100nm (Can be Customized) |
| Purity | 99.9% |
| Color | White |
| Molecular Weight | 60.08 g/ mol |
| Density | 2.6 g/ cm³ |
| Melting Point | 1713 ºC |
| Boiling Point | 2950 ºC |
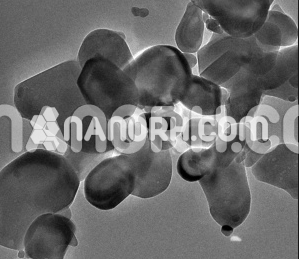
Silica Nanoparticles
Round silicon Oxide nanoparticles / SiO2 circular powder is generally utilized as a part of vast scale, ultra-expensive scale coordinated circuits, epoxy forming compound, electronic preparing, top of the line cosmetics? C precision earthenware production, natural glass, epoxy elastic, aviation pottery, and different businesses et cetera.
Application of silicon Oxide nanoparticles as fillers in the preparation of nanocomposite of polymers has drawnmuch attention, due to the increased demand for new materials with improved thermal, mechanical, physical, and chemical properties. Recent developments in the synthesis of monodispersed, narrow-size distribution of nanoparticles by sol-gel method provide significant boost to development of silica-polymer nanocomposites. This paper is written by emphasizing on the synthesis of silicon oxide nanoparticles, characterization on size-dependent properties, and surface modification for the preparation of homogeneous nanocomposites, generally by sol-gel technique. The effect of nanosilica on the properties of various types of silica-polymer composites is also summarized. Silicon oxide nanoparticles can also be produced through high temperature flame decomposition of metal-organic precursors. This process is also referred to as chemical vapour condensation (CVC). In a typical CVC process, silica nanoparticles are produced by reacting silicon tetrachloride, SiCl4 with hydrogen and oxygen. Difficulty in controlling the particle size, morphology, and phase composition is the main disadvantage of the flame synthesis. Nevertheless, this is the prominent method that has been used to commercially produce silica nanoparticles in powder form.