| Sn-Cu Alloy Nanopowder | |
| Product No | NRE- 2013 |
| CAS No. | 7440-02-0/7440-32-6 |
| Formula | Sn-Cu |
| APS | <100nm (Can be Customized) |
| Purity | 99.9% |
| Color | Grey Black |
| Molecular Weight | 182.25g/mol |
| Density | NA |
| Melting Point | NA |
| Boiling Point | NA |
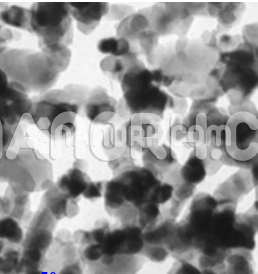
Sn-Cu Alloy Nanopowder
Sn – 0.8% by weight of copper alloy is prepared to study the transformation of white tin into the gray tin. The relationship between the transformation percentage and the time is determined by measuring the areas of the transformed gray can and examining the effect of cold rolling on the transformation. It is concluded that both copper additions to the pure tin and severe deformation are effective in improving transformation. The incubation period before the start of the transformation also depends on the reduction of the cold.
Three types of free ligands Sn-Cu alloys Nanopowder are prepared by electrodeposition, then distributed and electrochemically tested as possible materials for negative electrodes of lithium-ion batteries. The morphology and structure of these alloys have been characterized by X-ray diffraction, scanning electron microscopy, and atomic absorption spectrophotometer. Experimental results indicate that the composition, pore size, size distribution, and alloy homogeneity can be controlled easily by changing the proportion of primary salts in the plating bath. The content of Sn in the alloys after an electrochemical sale is significantly reduced and Cu6Sn5 becomes the main active component. However, there is no obvious decrease in a specific capacity. The initial specific capacities of all materials are in the range 480-810 mAh g-1. But the ability of the deposited Sn-Cu alloys remains 110-190 mAh g-1 after 50 cycles, while a capacity of Sn-Cu nanoporous alloys remains approximately 400 mAh g-1 after the same cycles. This suggests that both the capacity and performance of the cycle can be improved by optimizing the nanostructured materials using electrochemical distribution.