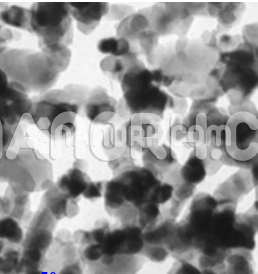
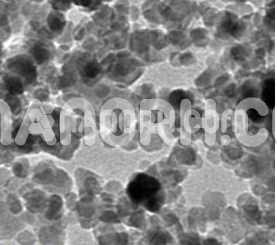
Yttrium Oxide Nanopowder / Nanoparticles (Y2O3, 99.999%, 20-40 nm)
Yttrium oxide is Y2O3. It is an air-steady, white strong substance. Yttrium oxide is utilized as a typical beginning material for the two materials science and additionally inorganic mixes. In materials science: It is the most vital yttrium compound and is broadly used to make YVO4 europium and Y2O3 europium phosphors that give the red shading in shading TV picture tubes….
| Yttrium Oxide Nanopowder | |
| Product No | NRE-3065 |
| CAS No. | 1314-36-9 |
| Formula | Y2O3 |
| APS | <40nm (Can be Customized) |
| Purity | 99.9999% |
| Color | white |
| Molecular Weight | 225.81 g/mol |
| Density | 5.01 g/cm3 |
| Melting Point | 2425 °C |
| Boiling Point | 4300 °C |
Yttrium Oxide Nanopowder (Y2O3) Applications:
In the field of materials science, these particles have numerous applications, such as giving color to television picture tubes. It is also used in the production of plasma screens and flat panels. Yttrium iron garnets derived from yttrium oxide are used as powerful microwave filters. Yttrium oxide is an important starting point in the inorganic synthesis of compounds. The property of emitting red light is used in the manufacture of fluorescent lamps. They are also used as additives in high temperature coatings, paints and plastics for UV protection and in the manufacture of permanent magnets. In ultrafast sensors used in G and X-rays. Some other uses include additives in steel, non-ferrous alloys and iron.
Yttrium oxide is Y2O3. It is an air-steady, white strong substance. Yttrium oxide is utilized as a typical beginning material for the two materials science and additionally inorganic mixes. In materials science: It is the most vital yttrium compound and is broadly used to make YVO4 europium and Y2O3 europium phosphors that give the red shading in shading TV picture tubes. Yttrium oxide is likewise used to influence yttrium to press garnets, which are exceptionally compelling microwave channels. In inorganic union: Yttrium oxide is an essential beginning stage for inorganic mixes. For organometallic science, it is changed over to YCl3 in response with concentrated hydrochloric corrosive and ammonium chloride. In different utilizations: Coatings for high-temperature applications; Display materials (with low-vitality excitation sources); Fluorescent for transmission electron microscopy; Ultrafast sensors (for x-beam, g-beam recognition and quick scintillator phosphor); Additives in paints and plastics for securing UV corruption; Additives in perpetual magnets; Red radiating materials in fluorescent lights; Additives in steel, press, and non-ferrous composites; Photoelectric (sun-powered cells) sensors; Plasma show boards; Getters; High-powder lasers for boring, cutting and welding; Infrared protecting covering; Flat-board shows; Dilutes for nuclear heap fuel; Cathode beam tube screens; Field-outflow shows; Engine parts; Dopants in SrZrO3…